
最新刊期
卷 51 , 期 1 , 2026
-
 摘要:Under the dual challenges of global warming and fossil energy depletion, CO2 hydrogenation to aromatics provides a green pathway for carbon resource recycling. CO2 hydrogenation to aromatics mainly focuses on two reaction pathways: CO2-modified Fischer-Tropsch synthesis (CO2-FTS) and methanol-mediated pathway. The reaction mechanisms and key intermediates of CO2-FTS and methanol-mediated pathways for the production of aromatics were reviewed, and the recent progress on catalyst structure design and optimization was summarized. The effects of additive type, support structure, acidity and structure modulation of zeolite molecular sieves, and the distance of active components in the bifunctional catalysts on the catalytic performance of CO2 hydrogenation to aromatics were highlighted. The synergistic effect of CO2 hydrogenation and arylation reaction as well as the formation of hydrogenation intermediates are the key factors influencing the performance of the catalysts. Finally, the application prospects of the catalysts for CO2 hydrogenation to aromatics were discussed, providing a reference for promoting the development in green chemical technology.关键词:CO2 hydrogenation;aromatics;bifunctional catalysts;reaction pathways0|0|0更新时间:2026-02-02
摘要:Under the dual challenges of global warming and fossil energy depletion, CO2 hydrogenation to aromatics provides a green pathway for carbon resource recycling. CO2 hydrogenation to aromatics mainly focuses on two reaction pathways: CO2-modified Fischer-Tropsch synthesis (CO2-FTS) and methanol-mediated pathway. The reaction mechanisms and key intermediates of CO2-FTS and methanol-mediated pathways for the production of aromatics were reviewed, and the recent progress on catalyst structure design and optimization was summarized. The effects of additive type, support structure, acidity and structure modulation of zeolite molecular sieves, and the distance of active components in the bifunctional catalysts on the catalytic performance of CO2 hydrogenation to aromatics were highlighted. The synergistic effect of CO2 hydrogenation and arylation reaction as well as the formation of hydrogenation intermediates are the key factors influencing the performance of the catalysts. Finally, the application prospects of the catalysts for CO2 hydrogenation to aromatics were discussed, providing a reference for promoting the development in green chemical technology.关键词:CO2 hydrogenation;aromatics;bifunctional catalysts;reaction pathways0|0|0更新时间:2026-02-02 -
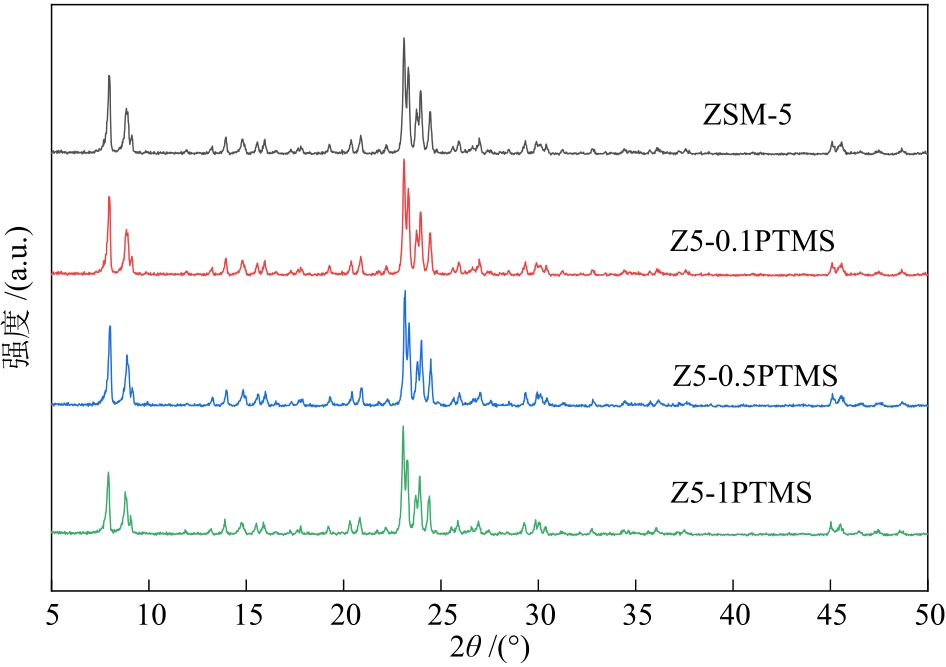 摘要:CO2 hydrogenation to sustainable aviation fuels can not only help achieve “carbon neutrality”, but also support the sustainable development of the aviation industry. However, due to the difficulty in activating CO2 as an inert molecule and the difficulty in generating sustainable aviation fuels (C8~C16 hydrocarbons) by C—C coupling, catalyst design has faced significant challenges. Propyl trimethoxysilane (PTMS) was used to modify ZSM-5 zeolites, and ZSM-5 zeolites with varying water contact angles were successfully prepared after hydrophobic modification. They were combined with Na-Fe3O4 to construct bifunctional catalysts for CO2 hydrogenation. The results show that propyl trimethoxysilane modification can not only enhance the hydrophobicity of ZSM-5 zeolites, but also inhibit the cracking of long-chain hydrocarbons on the surface of ZSM-5 zeolites to improve the sustainable aviation fuels selectivity by the acidic sites coverage on the outer surface of ZSM-5 zeolites with hydrophobic groups. Under the reaction conditions of 320 ℃, 3 MPa and n(H2):n(CO2) = 3:1, NaFe/Z5-0.5PTMS has the best catalytic performance for CO2 hydrogenation, with the CO2 conversion rate of 32.6%, CO selectivity of 9.9% and sustainable aviation fuels selectivity of 38.8%.关键词:CO2 hydrogenation;sustainable aviation fuels;hydrophobic modification;ZSM-579|0|0更新时间:2026-02-02
摘要:CO2 hydrogenation to sustainable aviation fuels can not only help achieve “carbon neutrality”, but also support the sustainable development of the aviation industry. However, due to the difficulty in activating CO2 as an inert molecule and the difficulty in generating sustainable aviation fuels (C8~C16 hydrocarbons) by C—C coupling, catalyst design has faced significant challenges. Propyl trimethoxysilane (PTMS) was used to modify ZSM-5 zeolites, and ZSM-5 zeolites with varying water contact angles were successfully prepared after hydrophobic modification. They were combined with Na-Fe3O4 to construct bifunctional catalysts for CO2 hydrogenation. The results show that propyl trimethoxysilane modification can not only enhance the hydrophobicity of ZSM-5 zeolites, but also inhibit the cracking of long-chain hydrocarbons on the surface of ZSM-5 zeolites to improve the sustainable aviation fuels selectivity by the acidic sites coverage on the outer surface of ZSM-5 zeolites with hydrophobic groups. Under the reaction conditions of 320 ℃, 3 MPa and n(H2):n(CO2) = 3:1, NaFe/Z5-0.5PTMS has the best catalytic performance for CO2 hydrogenation, with the CO2 conversion rate of 32.6%, CO selectivity of 9.9% and sustainable aviation fuels selectivity of 38.8%.关键词:CO2 hydrogenation;sustainable aviation fuels;hydrophobic modification;ZSM-579|0|0更新时间:2026-02-02 -
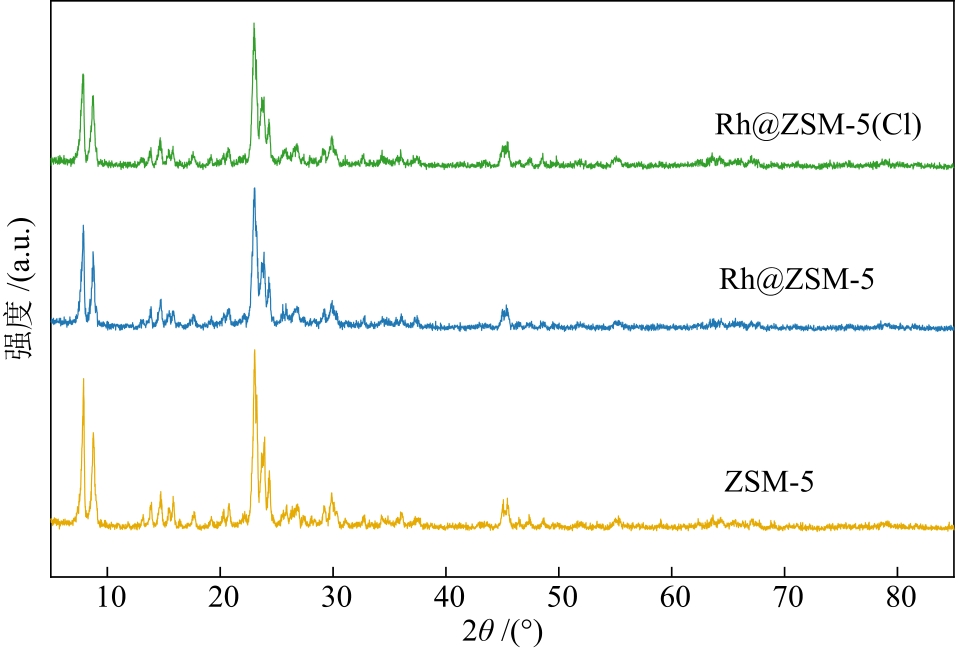 摘要:Methane (CH4) is a “double-edged sword” energy source with high calorific value and strong greenhouse effect, so the development of catalytic conversion technology for CH4 is crucial for energy utilization and environmental protection. Although noble metal catalysts show high CH4 conversion rates and product selectivities in partial oxidation of methane (POM) reaction, they face the problem of high cost. It is a feasible cost reduction method to improve the utilization rate of noble metals by increasing the dispersion of active metals through the domain-limiting effect of molecular sieves. In order to further improve the catalytic performances of catalysts for POM reaction, a hydrothermal synthesis method was used to prepare Rh@ZSM-5(Cl) and Rh@ZSM-5 catalysts with rhodium chloride and rhodium nitrate as precursors, respectively. The catalysts and ZSM-5 support were characterized by XRD, TEM, N2 adsorption/desorption and so on. The regulatory effect of precursor introduction on the catalytic performances of catalysts were explored, and the differences of catalytic performances were tested. The results show that rhodium chloride precursor induces the generation of O—Rh—Cl active species, significantly enhancing the interaction between active metals and the carriers. Rh@ZSM-5(Cl) exhibits excellent catalytic performance for POM reaction, with CH4 conversion rate of 90.7% and H2 selectivity of 94.5% at 900 ℃, attributed to the dynamic coupling of water-gas shift reaction (WGSR) and POM reaction promoted by O—Rh—Cl active species.关键词:partial oxidation of methane reaction;Rh;ZSM-5 zeolite;syngas32|0|0更新时间:2026-02-02
摘要:Methane (CH4) is a “double-edged sword” energy source with high calorific value and strong greenhouse effect, so the development of catalytic conversion technology for CH4 is crucial for energy utilization and environmental protection. Although noble metal catalysts show high CH4 conversion rates and product selectivities in partial oxidation of methane (POM) reaction, they face the problem of high cost. It is a feasible cost reduction method to improve the utilization rate of noble metals by increasing the dispersion of active metals through the domain-limiting effect of molecular sieves. In order to further improve the catalytic performances of catalysts for POM reaction, a hydrothermal synthesis method was used to prepare Rh@ZSM-5(Cl) and Rh@ZSM-5 catalysts with rhodium chloride and rhodium nitrate as precursors, respectively. The catalysts and ZSM-5 support were characterized by XRD, TEM, N2 adsorption/desorption and so on. The regulatory effect of precursor introduction on the catalytic performances of catalysts were explored, and the differences of catalytic performances were tested. The results show that rhodium chloride precursor induces the generation of O—Rh—Cl active species, significantly enhancing the interaction between active metals and the carriers. Rh@ZSM-5(Cl) exhibits excellent catalytic performance for POM reaction, with CH4 conversion rate of 90.7% and H2 selectivity of 94.5% at 900 ℃, attributed to the dynamic coupling of water-gas shift reaction (WGSR) and POM reaction promoted by O—Rh—Cl active species.关键词:partial oxidation of methane reaction;Rh;ZSM-5 zeolite;syngas32|0|0更新时间:2026-02-02 -
 摘要:Methane dehydroaromatization (MDA) represents a promising non-petroleum route for aromatic production. Exploring the effects of the pore structures of catalysts on their MDA reaction catalytic performances is crucial for developing high-efficiency catalysts. MWW-type zeolites (MCM-56 and MCM-22) were synthesized by seed-assisted method. The phases, morphologies and pore structures of Mo/MCM-56 and Mo/MCM-22 catalysts were characterized by XRD, TEM, SEM, and N2 adsorption/desorption. Furthermore, the effects of different pore channel systems of MWW-type zeolites on catalytic performances of Mo/MCM-56 and Mo/MCM-22 catalysts for MDA reaction were investigated. The results show that the seed-assisted method significantly reduces crystallization time of zeolites, and the structures of MWW zeolites are closely related to the structures of seeds. After reacting for 9.0 h under the same conditions (700 °C, 100 kPa), Mo/MCM-22 demonstrates higher aromatic selectivity (around 95%) and aromatic yield (around 7.5%), and superior catalytic stability compared to Mo/MCM-56. Therefore, it is speculated that the 12-membered ring supercages, formed through condensation of adjacent two-dimensional MWW zeolite nanosheets in Mo/MCM-22 catalyst, serves as the primary sites for MDA reaction. The study reveals that different pore channel systems of MWW zeolites have significant effects on the catalytic performance of catalysts for MDA reaction, providing new ideas for designing highly selective and stable MDA catalysts.关键词:methane dehydroaromatization;MWW-type zeolites;catalysts;seed-assisted method24|0|0更新时间:2026-02-02
摘要:Methane dehydroaromatization (MDA) represents a promising non-petroleum route for aromatic production. Exploring the effects of the pore structures of catalysts on their MDA reaction catalytic performances is crucial for developing high-efficiency catalysts. MWW-type zeolites (MCM-56 and MCM-22) were synthesized by seed-assisted method. The phases, morphologies and pore structures of Mo/MCM-56 and Mo/MCM-22 catalysts were characterized by XRD, TEM, SEM, and N2 adsorption/desorption. Furthermore, the effects of different pore channel systems of MWW-type zeolites on catalytic performances of Mo/MCM-56 and Mo/MCM-22 catalysts for MDA reaction were investigated. The results show that the seed-assisted method significantly reduces crystallization time of zeolites, and the structures of MWW zeolites are closely related to the structures of seeds. After reacting for 9.0 h under the same conditions (700 °C, 100 kPa), Mo/MCM-22 demonstrates higher aromatic selectivity (around 95%) and aromatic yield (around 7.5%), and superior catalytic stability compared to Mo/MCM-56. Therefore, it is speculated that the 12-membered ring supercages, formed through condensation of adjacent two-dimensional MWW zeolite nanosheets in Mo/MCM-22 catalyst, serves as the primary sites for MDA reaction. The study reveals that different pore channel systems of MWW zeolites have significant effects on the catalytic performance of catalysts for MDA reaction, providing new ideas for designing highly selective and stable MDA catalysts.关键词:methane dehydroaromatization;MWW-type zeolites;catalysts;seed-assisted method24|0|0更新时间:2026-02-02 -
 摘要:As a crucial basic chemical raw material, propylene holds significant strategic importance in chemical industry. With the rapid expansion of propylene downstream industrial chains and the feedstock advantages brought by shale gas revolution, propane direct dehydrogenation (PDH) process has emerged as one of the mainstream technical routes for propylene production due to its substantially reduced manufacturing costs. In this context, developing high-efficiency chromium-based catalysts with integrated high catalytic activity, product selectivity and stability has become a key research focus in this field. According to the research status of PDH chromium-based catalysts, the PDH reaction mechanism and the catalyst deactivation mechanism under chromium-based catalysts were reviewed. The influences of important components of PDH chromium-based catalysts, such as active centers, supports and promoters on their catalytic performance were analyzed. And various preparation methods and improvement strategies for PDH chromium-based catalysts were summarized. Finally, the future development directions of PDH chromium-based catalysts were prospected.关键词:propylene;propane direct dehydrogenation;chromium-based catalysts;active centers57|0|0更新时间:2026-02-02
摘要:As a crucial basic chemical raw material, propylene holds significant strategic importance in chemical industry. With the rapid expansion of propylene downstream industrial chains and the feedstock advantages brought by shale gas revolution, propane direct dehydrogenation (PDH) process has emerged as one of the mainstream technical routes for propylene production due to its substantially reduced manufacturing costs. In this context, developing high-efficiency chromium-based catalysts with integrated high catalytic activity, product selectivity and stability has become a key research focus in this field. According to the research status of PDH chromium-based catalysts, the PDH reaction mechanism and the catalyst deactivation mechanism under chromium-based catalysts were reviewed. The influences of important components of PDH chromium-based catalysts, such as active centers, supports and promoters on their catalytic performance were analyzed. And various preparation methods and improvement strategies for PDH chromium-based catalysts were summarized. Finally, the future development directions of PDH chromium-based catalysts were prospected.关键词:propylene;propane direct dehydrogenation;chromium-based catalysts;active centers57|0|0更新时间:2026-02-02 -
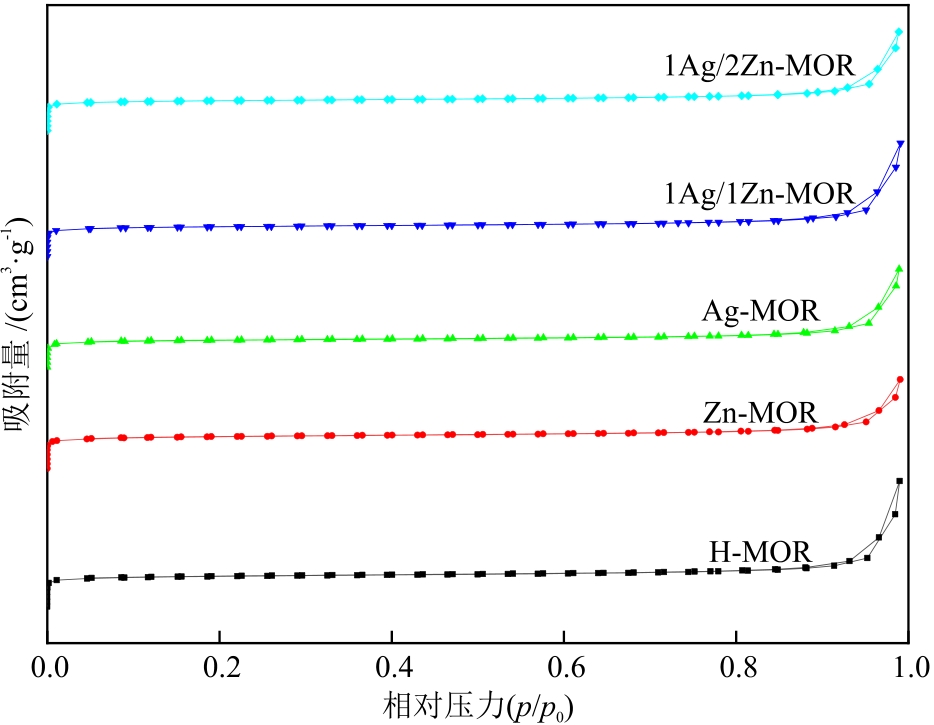 摘要:In response to the problem of easy deactivation of mordenite (MOR) catalyst in the dimethyl ether (DME) carbonylation reaction, the ion-exchange method was adopted to introduce Zn and Ag bimetals for the modification of MOR to optimize the distribution of acidic sites and the pore structure so as to enhance the catalytic performance. A series of catalysts were prepared by hydrothermal synthesis method, and their physicochemical properties were analyzed by characterization techniques, such as XRD, ICP-AES, SEM, NH3-TPD and Py-IR. The catalytic activity was evaluated in a fixed bed reactor. The results show that after the introduction of Ag, both the amount and strength of Brønsted acid in eight-membered ring channels of the catalyst increase significantly. After the introduction of Zn, the amount of Lewis acid in the twelve-membered ring channels increases, which synergistically promotes the adsorption of CO and the reaction kinetics. Under the conditions of 200 ℃, 1.0 MPa and space velocity of 2400 mL/(g·h), the 1Ag/2Zn-MOR catalyst demonstrates the optimal catalytic performance. After reaction for 3 h with the catalyst, the conversion rate of DME reaches 98%. Even after reaction for 25 h, the DME conversion rate still remains 90%, and the selectivity of methyl acetate is always greater than 99%. The modification with Zn-Ag bimetals can effectively enhance the catalytic activity and stability by regulating the distribution of acid sites and the pore structure of the catalyst, which provides a theoretical foundation for the development of highly efficient catalysts for DME carbonylation.关键词:dimethyl ether carbonylation;hydrothermal synthesis;ion-exchange method;mordenite;bimetallic modification41|0|0更新时间:2026-02-02
摘要:In response to the problem of easy deactivation of mordenite (MOR) catalyst in the dimethyl ether (DME) carbonylation reaction, the ion-exchange method was adopted to introduce Zn and Ag bimetals for the modification of MOR to optimize the distribution of acidic sites and the pore structure so as to enhance the catalytic performance. A series of catalysts were prepared by hydrothermal synthesis method, and their physicochemical properties were analyzed by characterization techniques, such as XRD, ICP-AES, SEM, NH3-TPD and Py-IR. The catalytic activity was evaluated in a fixed bed reactor. The results show that after the introduction of Ag, both the amount and strength of Brønsted acid in eight-membered ring channels of the catalyst increase significantly. After the introduction of Zn, the amount of Lewis acid in the twelve-membered ring channels increases, which synergistically promotes the adsorption of CO and the reaction kinetics. Under the conditions of 200 ℃, 1.0 MPa and space velocity of 2400 mL/(g·h), the 1Ag/2Zn-MOR catalyst demonstrates the optimal catalytic performance. After reaction for 3 h with the catalyst, the conversion rate of DME reaches 98%. Even after reaction for 25 h, the DME conversion rate still remains 90%, and the selectivity of methyl acetate is always greater than 99%. The modification with Zn-Ag bimetals can effectively enhance the catalytic activity and stability by regulating the distribution of acid sites and the pore structure of the catalyst, which provides a theoretical foundation for the development of highly efficient catalysts for DME carbonylation.关键词:dimethyl ether carbonylation;hydrothermal synthesis;ion-exchange method;mordenite;bimetallic modification41|0|0更新时间:2026-02-02 -
 摘要:2-hydroxytetrahydrofuran (2-HTHF) is an important fine chemical material. The key to 2-HTHF production via hydroisomerization of cis-1,4-butenediol (cis-BED) lies in constructing the bifunctional catalyst with controllable metal center and acid strength. A series of Ni/WO3-ZrO2 bifunctional catalysts were prepared by incipient wetness impregnation method and their structures were characterized by NH3-TPD, XPS, XRD, etc. The effects of reduction temperatures, reduction time, loading amounts of Ni and WO3 and dispersions of Ni and WO3 on the structures and catalytic performances of catalysts were investigated. Additionally, the kinetics of the cis-BED hydroisomerization reaction over the optimal catalyst under specified conditions were studied. The results show that under the conditions of catalyst dosage of 0.2 g, reaction temperature of 200 ℃, H2 pressure of 0.4 MPa and gas hourly space velocity of 37.8 L/(g·h), 10%Ni/20%WO3-ZrO2-HT (loading amounts of Ni and WO3 are 10% and 20%, respectively) obtained by reducing at 600 ℃ for 2 h exhibits the best catalytic performance with cis-BED conversion rate of 67.9% and 2-HTHF selectivity of 30.1%, respectively. The established reaction network and kinetic model can effectively describ the cis-BED hydroisomerization process catalyzed by 10%Ni/20%WO3-ZrO2-HT, revealing that the primary pathway for 2-HTHF formation involves the isomerization of cis-BED to trans-1,4-butenediol, follows by its further conversion to 2-HTHF.关键词:1,4-butenediol;2-hydroxytetrahydrofuran;bifunctional catalysts;hydroisomerization;reaction kinetics39|0|0更新时间:2026-02-02
摘要:2-hydroxytetrahydrofuran (2-HTHF) is an important fine chemical material. The key to 2-HTHF production via hydroisomerization of cis-1,4-butenediol (cis-BED) lies in constructing the bifunctional catalyst with controllable metal center and acid strength. A series of Ni/WO3-ZrO2 bifunctional catalysts were prepared by incipient wetness impregnation method and their structures were characterized by NH3-TPD, XPS, XRD, etc. The effects of reduction temperatures, reduction time, loading amounts of Ni and WO3 and dispersions of Ni and WO3 on the structures and catalytic performances of catalysts were investigated. Additionally, the kinetics of the cis-BED hydroisomerization reaction over the optimal catalyst under specified conditions were studied. The results show that under the conditions of catalyst dosage of 0.2 g, reaction temperature of 200 ℃, H2 pressure of 0.4 MPa and gas hourly space velocity of 37.8 L/(g·h), 10%Ni/20%WO3-ZrO2-HT (loading amounts of Ni and WO3 are 10% and 20%, respectively) obtained by reducing at 600 ℃ for 2 h exhibits the best catalytic performance with cis-BED conversion rate of 67.9% and 2-HTHF selectivity of 30.1%, respectively. The established reaction network and kinetic model can effectively describ the cis-BED hydroisomerization process catalyzed by 10%Ni/20%WO3-ZrO2-HT, revealing that the primary pathway for 2-HTHF formation involves the isomerization of cis-BED to trans-1,4-butenediol, follows by its further conversion to 2-HTHF.关键词:1,4-butenediol;2-hydroxytetrahydrofuran;bifunctional catalysts;hydroisomerization;reaction kinetics39|0|0更新时间:2026-02-02 -
 摘要:Solar-driven interfacial evaporation (SIE) technology, with its advantages of environmental friendliness and low energy consumption, offers a novel solution to the global challenges of water scarcity and energy crisis. In recent years, carbon-based materials have demonstrated great application potential in this field due to their excellent photothermal conversion performance, large specific surface area and good chemical stability. Carbon-based materials can significantly enhance energy conversion efficiency, reduce system costs, and play a role in reducing carbon emissions. The research progress of carbon-based materials in solar-driven interfacial evaporation technology was summarized, with a focus on in-depth discussion of their photothermal conversion, water transport and anti-salting mechanisms. Their potential for carbon emission reduction was analyzed, and their application prospects in seawater desalination and wastewater treatment were prospected, aming to provide a reference for the development of efficient, low-cost and sustainable water treatment technologies, contributing to the realization of “carbon neutrality” goals.关键词:solar-driven interfacial evaporation;carbon-based materials;carbon emission reduction;water treatment259|0|0更新时间:2026-02-02
摘要:Solar-driven interfacial evaporation (SIE) technology, with its advantages of environmental friendliness and low energy consumption, offers a novel solution to the global challenges of water scarcity and energy crisis. In recent years, carbon-based materials have demonstrated great application potential in this field due to their excellent photothermal conversion performance, large specific surface area and good chemical stability. Carbon-based materials can significantly enhance energy conversion efficiency, reduce system costs, and play a role in reducing carbon emissions. The research progress of carbon-based materials in solar-driven interfacial evaporation technology was summarized, with a focus on in-depth discussion of their photothermal conversion, water transport and anti-salting mechanisms. Their potential for carbon emission reduction was analyzed, and their application prospects in seawater desalination and wastewater treatment were prospected, aming to provide a reference for the development of efficient, low-cost and sustainable water treatment technologies, contributing to the realization of “carbon neutrality” goals.关键词:solar-driven interfacial evaporation;carbon-based materials;carbon emission reduction;water treatment259|0|0更新时间:2026-02-02 -
 摘要:MXene, as a new type of two-dimensional transition metal carbide/nitride material, has shown excellent performance in the field of separation and purification owing to its unique structure and abundant surface functional groups. Based on relevant theoretical and experimental research results, the physicochemical properties of MXene membranes were introduced, including post-treatment stability, electrical properties and mechanical properties, and the research progress on their applications in gas separation and capture, ion sieving, water purification and organic solvent purification was reviewed. Although MXene membranes exhibit great application potential in the field of separation and purification, they still face challenges such as stability, interlayer spacing regulation, swelling, interfacial compatibility, cost and environmental safety. Future research needs to integrate strategies such as surface modification, structural design and process optimization, while exploring low-cost preparation techniques and strengthening environmental safety assessments, so as to promote the application of MXene membranes in broader fields.关键词:membrane separation;two-dimensional MXene membranes;gas separation;ion sieving;water purification;organic solvent purification71|0|0更新时间:2026-02-02
摘要:MXene, as a new type of two-dimensional transition metal carbide/nitride material, has shown excellent performance in the field of separation and purification owing to its unique structure and abundant surface functional groups. Based on relevant theoretical and experimental research results, the physicochemical properties of MXene membranes were introduced, including post-treatment stability, electrical properties and mechanical properties, and the research progress on their applications in gas separation and capture, ion sieving, water purification and organic solvent purification was reviewed. Although MXene membranes exhibit great application potential in the field of separation and purification, they still face challenges such as stability, interlayer spacing regulation, swelling, interfacial compatibility, cost and environmental safety. Future research needs to integrate strategies such as surface modification, structural design and process optimization, while exploring low-cost preparation techniques and strengthening environmental safety assessments, so as to promote the application of MXene membranes in broader fields.关键词:membrane separation;two-dimensional MXene membranes;gas separation;ion sieving;water purification;organic solvent purification71|0|0更新时间:2026-02-02 -
 摘要:COS hydrolysis catalysis is important in industrial gas purification and desulfurization. MgAl hydrotalcite-based oxides (MgAl-MMO) can effectively catalyze COS hydrolysis in a simple atmosphere (COS + H2O). However, the catalytic effect of O2/H2S on MgAl-MMO is not clear, and the existing synthesis methods are time-consuming. The feasibility of microwave-assisted synthesis of MgAl-MMO was explored and its catalytic behavior under reaction gas containing O2/H2S was investigated. The results show that the best performance of the catalyst for COS hydrolysis is achieved by microwave crystallization at 80 ℃ and n(Mg)/n(Al) of 2. The COS conversion rate and H2S selectivity of the catalysts are 100.0% and maintain for 540 min in in the reaction gas (O2 volume fraction of 1.0%). However, the COS conversion rate and H2S selectivity decrease significantly (by 63.2% and 68.8%, respectively) when the O2 volume fraction increases to 1.5%, which is mainly attributed to the increase of sulfur deposition on the catalyst surface due to the catalytic oxidation of H2S at high O2 volume fraction. H2S in the O2-free environment leads to catalyst poisoning during the catalytic process, and the degree of poisoning increases with the increase of H2S mass concentration: COS conversion rate and H2S selectivity with 198 mg/m3 H2S decrease to 37.4% and 5.3% after 100 mintutes, respectively. The coexistence of 1.0% O2 and 198 mg/m3 H2S further accelerate the catalyst deactivation, exacerbate the surface sulfation and the relative content of medium bases and weak bases decreased significantly, and the detrimental effect of H2S on the catalyst is significantly greater than that of O2, with the COS conversion rate and the H2S selectivity of only 20.2% and 1.4%, respectively, after 85 minutes of reaction.关键词:microwave synthesis;MgAl hydrotalcite-based oxides;COS hydrolysis;H2S;deactivation27|0|0更新时间:2026-02-02
摘要:COS hydrolysis catalysis is important in industrial gas purification and desulfurization. MgAl hydrotalcite-based oxides (MgAl-MMO) can effectively catalyze COS hydrolysis in a simple atmosphere (COS + H2O). However, the catalytic effect of O2/H2S on MgAl-MMO is not clear, and the existing synthesis methods are time-consuming. The feasibility of microwave-assisted synthesis of MgAl-MMO was explored and its catalytic behavior under reaction gas containing O2/H2S was investigated. The results show that the best performance of the catalyst for COS hydrolysis is achieved by microwave crystallization at 80 ℃ and n(Mg)/n(Al) of 2. The COS conversion rate and H2S selectivity of the catalysts are 100.0% and maintain for 540 min in in the reaction gas (O2 volume fraction of 1.0%). However, the COS conversion rate and H2S selectivity decrease significantly (by 63.2% and 68.8%, respectively) when the O2 volume fraction increases to 1.5%, which is mainly attributed to the increase of sulfur deposition on the catalyst surface due to the catalytic oxidation of H2S at high O2 volume fraction. H2S in the O2-free environment leads to catalyst poisoning during the catalytic process, and the degree of poisoning increases with the increase of H2S mass concentration: COS conversion rate and H2S selectivity with 198 mg/m3 H2S decrease to 37.4% and 5.3% after 100 mintutes, respectively. The coexistence of 1.0% O2 and 198 mg/m3 H2S further accelerate the catalyst deactivation, exacerbate the surface sulfation and the relative content of medium bases and weak bases decreased significantly, and the detrimental effect of H2S on the catalyst is significantly greater than that of O2, with the COS conversion rate and the H2S selectivity of only 20.2% and 1.4%, respectively, after 85 minutes of reaction.关键词:microwave synthesis;MgAl hydrotalcite-based oxides;COS hydrolysis;H2S;deactivation27|0|0更新时间:2026-02-02 -
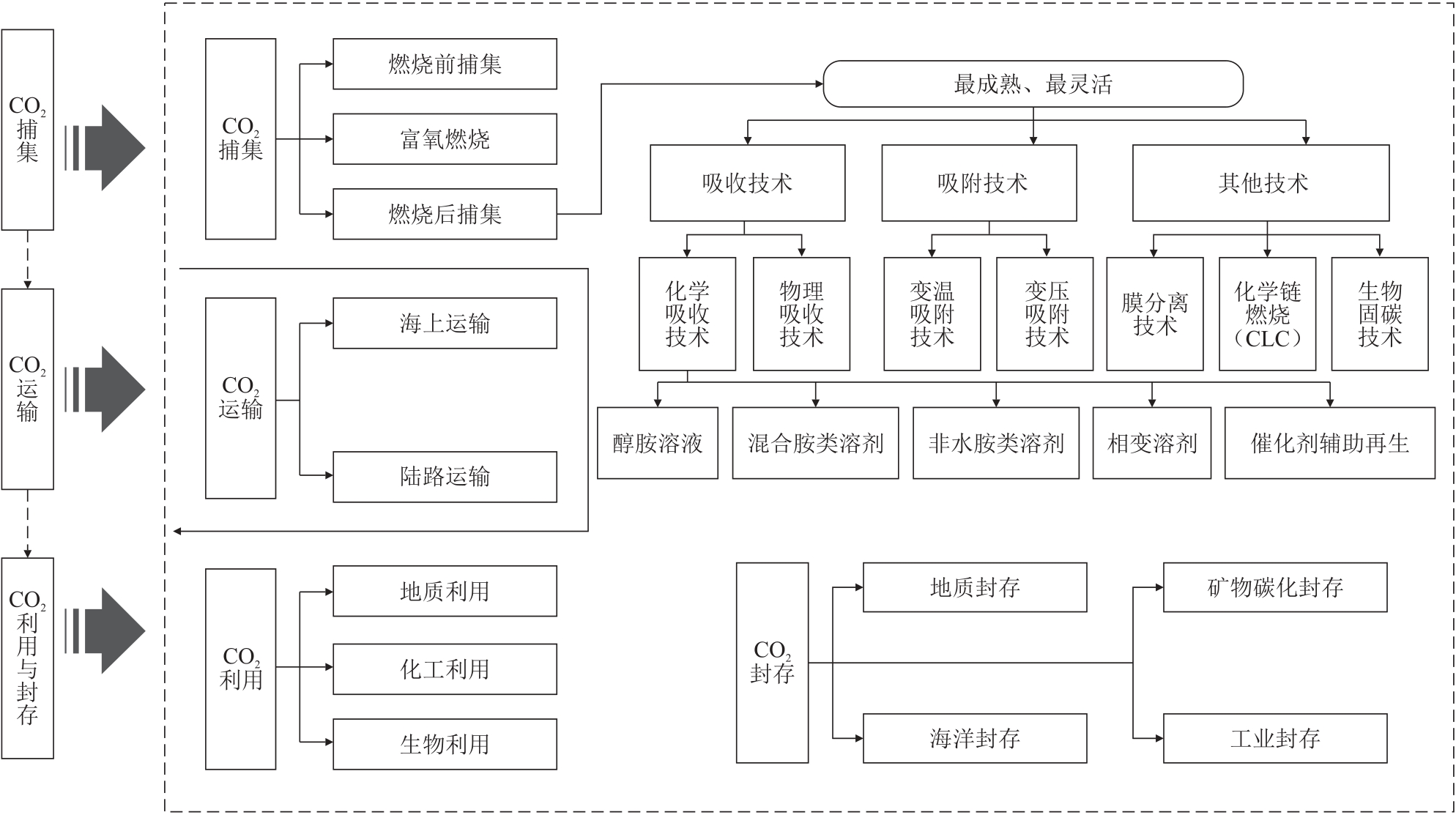 摘要:Carbon capture, utilization, and storage (CCUS) technology represents a critical pathway for achieving low-carbon transformation in the power industries. Based on existing engineering practices, the current development status of CCUS was explored, and the characteristics and engineering application status of mainstream carbon capture technologies were analyzed, and the compatibility advantages of post-combustion capture technology with existing power plants were pointed out, and the matching relationships between different transportation modes and carbon capture scales were compared, and the supportive role of pipeline network transportation in large-scale applications was revealed, and the emission reduction potential of CO2 storage and utilization pathways was evaluated, and the development trend of CCUS was prospected. It is found that post-combustion capture technology is mature and has good inheritance to existing facilities, and urgent development of novel materials (absorbents/adsorbents/membranes) and process optimization to achieve low-energy consumption are required. Small-scale land transportation has achieved initial scale, while pipeline transportation is entering rapid development phase. Transportation costs have significantly decreased with the expansion of transportation scale, which can provide important guarantees for future industrial-scale CO2 transportation. CO2 flooding increases crude oil recovery rates by 7%~15%, and its combination with chemical/biological utilization is an effective measure to improve CCUS economic viability. Although current CCUS technology faces challenges of high energy consumption and cost, technological optimization and scaled applications can substantially enhance its economy. CCUS with bio-energy with carbon capture and storage (BECCS) technology is projected to contribute 10 × 108 t/a CO2 emission reduction for China by 2060, which can not only support deep decarbonization in the power industries but also provide technical references for low-carbon transitions in other high CO2 emission industries.关键词:CCUS;carbon capture;CO2 utilization and storage;power industries;engineering practice226|0|0更新时间:2026-02-02
摘要:Carbon capture, utilization, and storage (CCUS) technology represents a critical pathway for achieving low-carbon transformation in the power industries. Based on existing engineering practices, the current development status of CCUS was explored, and the characteristics and engineering application status of mainstream carbon capture technologies were analyzed, and the compatibility advantages of post-combustion capture technology with existing power plants were pointed out, and the matching relationships between different transportation modes and carbon capture scales were compared, and the supportive role of pipeline network transportation in large-scale applications was revealed, and the emission reduction potential of CO2 storage and utilization pathways was evaluated, and the development trend of CCUS was prospected. It is found that post-combustion capture technology is mature and has good inheritance to existing facilities, and urgent development of novel materials (absorbents/adsorbents/membranes) and process optimization to achieve low-energy consumption are required. Small-scale land transportation has achieved initial scale, while pipeline transportation is entering rapid development phase. Transportation costs have significantly decreased with the expansion of transportation scale, which can provide important guarantees for future industrial-scale CO2 transportation. CO2 flooding increases crude oil recovery rates by 7%~15%, and its combination with chemical/biological utilization is an effective measure to improve CCUS economic viability. Although current CCUS technology faces challenges of high energy consumption and cost, technological optimization and scaled applications can substantially enhance its economy. CCUS with bio-energy with carbon capture and storage (BECCS) technology is projected to contribute 10 × 108 t/a CO2 emission reduction for China by 2060, which can not only support deep decarbonization in the power industries but also provide technical references for low-carbon transitions in other high CO2 emission industries.关键词:CCUS;carbon capture;CO2 utilization and storage;power industries;engineering practice226|0|0更新时间:2026-02-02 -
 摘要:Carrying out CO2 capture is an effective approach to mitigating the contradiction between fossil energy utilization and global warming. Amine-based absorbents are the primary materials used for CO2 capture via chemical absorption. The mechanism of CO2 capture by amine-based absorbents was introduced and the structural characteristics and capture performance of various types were compared. Single-component amine absorbents feature simple preparation processes but suffer from low CO2 absorption capacity (from 0.50 mol/mol to 0.53 mol/mol) and high regeneration energy consumption (from 3.5 GJ/t to 4.0 GJ/t). By introducing other absorbents to form mixed amine systems, the CO2 absorption capacity can be increased to 0.706 mol/mol (2-amino-2-methyl-1-propanol and N-methyl-4-piperidinol), while reducing regeneration energy consumption and enhancing economic efficiency. Two-phase absorbents require heating only the CO2-rich phase, resulting in a regeneration energy consumption as low as 1.61 GJ/t (2-amino-2-methyl-1-propanol/piperazine/ethylene glycol dimethyl ether). They also exhibit good stability and thus show promising development prospects. Low-water (or anhydrous) absorbents exhibit outstanding CO2 absorption capacity but slower absorption rates, with regeneration energy consumption reaching 2.365 GJ/t (diethylenetriamine-triethanolamine). Novel absorbents such as ionic liquids show excellent CO2 capture performance and can effectively overcome problems associated with traditional absorbents, such as high energy consumption, rapid degradation, equipment corrosion and high cost. However, further research and validation are still required before large-scale application. The review can provide a reference for the selection and performance improvement of amine-based absorbents for CO2 capture.关键词:CO2 capture;amine-based absorbents;chemical absorption method;absorption performance;regeneration energy consumption4|0|0更新时间:2026-02-02
摘要:Carrying out CO2 capture is an effective approach to mitigating the contradiction between fossil energy utilization and global warming. Amine-based absorbents are the primary materials used for CO2 capture via chemical absorption. The mechanism of CO2 capture by amine-based absorbents was introduced and the structural characteristics and capture performance of various types were compared. Single-component amine absorbents feature simple preparation processes but suffer from low CO2 absorption capacity (from 0.50 mol/mol to 0.53 mol/mol) and high regeneration energy consumption (from 3.5 GJ/t to 4.0 GJ/t). By introducing other absorbents to form mixed amine systems, the CO2 absorption capacity can be increased to 0.706 mol/mol (2-amino-2-methyl-1-propanol and N-methyl-4-piperidinol), while reducing regeneration energy consumption and enhancing economic efficiency. Two-phase absorbents require heating only the CO2-rich phase, resulting in a regeneration energy consumption as low as 1.61 GJ/t (2-amino-2-methyl-1-propanol/piperazine/ethylene glycol dimethyl ether). They also exhibit good stability and thus show promising development prospects. Low-water (or anhydrous) absorbents exhibit outstanding CO2 absorption capacity but slower absorption rates, with regeneration energy consumption reaching 2.365 GJ/t (diethylenetriamine-triethanolamine). Novel absorbents such as ionic liquids show excellent CO2 capture performance and can effectively overcome problems associated with traditional absorbents, such as high energy consumption, rapid degradation, equipment corrosion and high cost. However, further research and validation are still required before large-scale application. The review can provide a reference for the selection and performance improvement of amine-based absorbents for CO2 capture.关键词:CO2 capture;amine-based absorbents;chemical absorption method;absorption performance;regeneration energy consumption4|0|0更新时间:2026-02-02 -
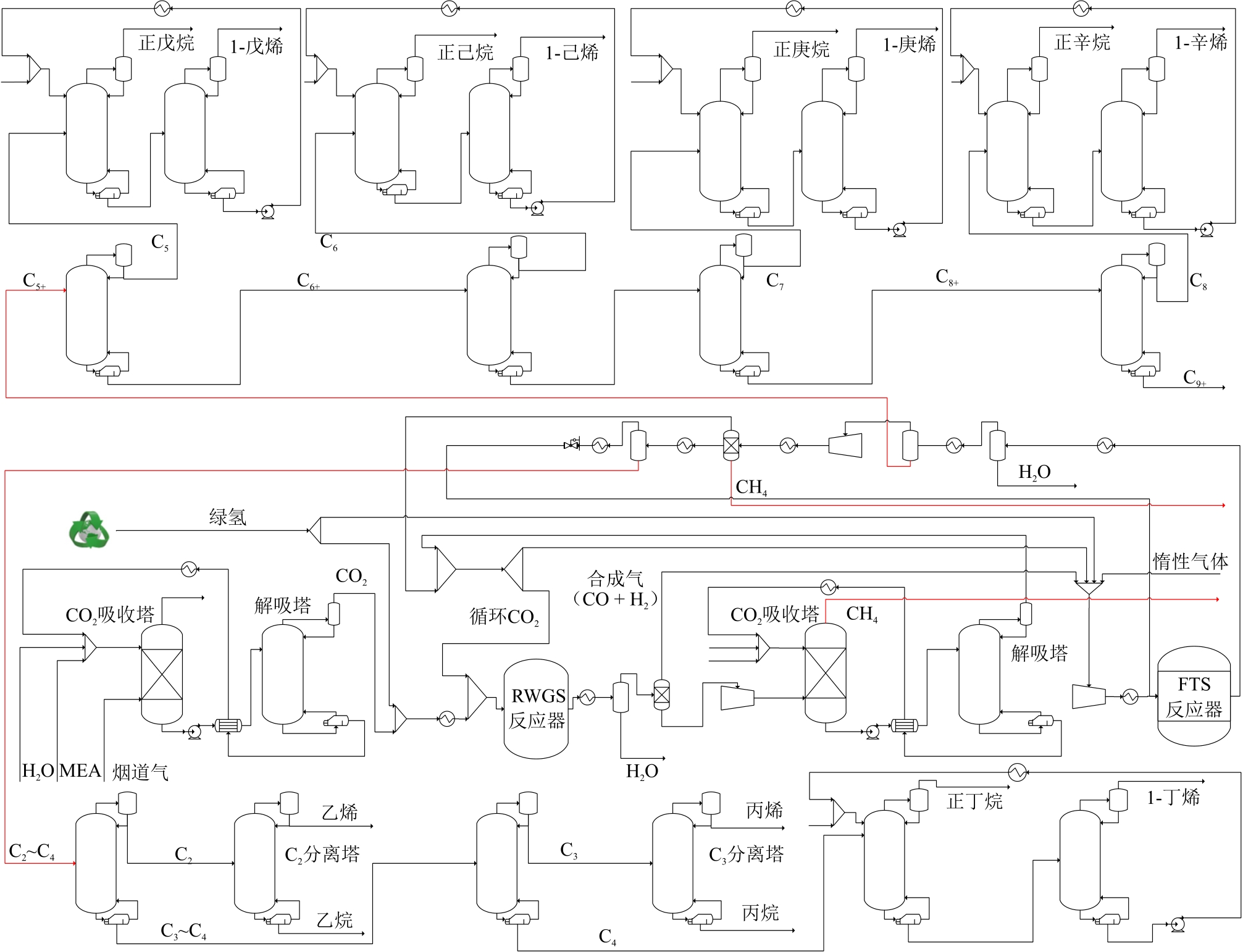 摘要:With the transition of the national energy structure and the advancement of “carbon peaking and carbon neutrality” strategy, CO2 hydrogenation to olefins process has become one of the key methods for carbon emission reduction and carbon utilization. Although experimental studies on catalysts for CO2 hydrogenation to olefins are relatively mature, their industrial application remains at a preliminary stage. Analyzing the process through simulation is one of the effective approaches to promoting their industrialization. Based on the catalysts for CO2 hydrogenation to olefins, a coupled process for converting CO2 from coal-fired flue gas and green H2 to olefins (“coupling process” for short) has been established for olefin production capacity of 70 × 104 t/a. The process encompasses multiple units, including carbon capture, the reverse water-gas shift reaction, Fischer-Tropsch synthesis to olefins and the subsequent separation of light and heavy hydrocarbons. Simulation results indicate that the coupling process offers significant carbon reduction benefits, achieving the carbon efficiency of 99.71% and net CO2 emission of -1.83 t/t (the CO2 emission for production 1 t olefins is -1.83 t). Enhancing CO2 conversion rate can effectively reduce capital investment and improve energy efficiency, while the cost of H2 production plays a critical role in determining the economic viability of the coupled process. When the CO2 conversion rate increases from 47% to 79%, the energy efficiency of the coupling process reaches 40.92% and the total capital investment can be reduced by 38.79%. When the cost of H2 production decreases from 0.367 USD/m3 (standard conditions) to 0.114 USD/m3, the production cost (refers to “olefins production cost”) of the coupled process can be reduced by 51.58%, reaching as low as 1006.1 USD/t. At the similar H2 production cost (0.317 USD/m3), the production cost of the process is 2100.8 USD/t, which is lower than the reported value (3580 USD/t) in literature.关键词:CO2 hydrogenation;green olefins;Aspen Plus stimulation;process feasibility analysis;economic analysis100|0|0更新时间:2026-02-02
摘要:With the transition of the national energy structure and the advancement of “carbon peaking and carbon neutrality” strategy, CO2 hydrogenation to olefins process has become one of the key methods for carbon emission reduction and carbon utilization. Although experimental studies on catalysts for CO2 hydrogenation to olefins are relatively mature, their industrial application remains at a preliminary stage. Analyzing the process through simulation is one of the effective approaches to promoting their industrialization. Based on the catalysts for CO2 hydrogenation to olefins, a coupled process for converting CO2 from coal-fired flue gas and green H2 to olefins (“coupling process” for short) has been established for olefin production capacity of 70 × 104 t/a. The process encompasses multiple units, including carbon capture, the reverse water-gas shift reaction, Fischer-Tropsch synthesis to olefins and the subsequent separation of light and heavy hydrocarbons. Simulation results indicate that the coupling process offers significant carbon reduction benefits, achieving the carbon efficiency of 99.71% and net CO2 emission of -1.83 t/t (the CO2 emission for production 1 t olefins is -1.83 t). Enhancing CO2 conversion rate can effectively reduce capital investment and improve energy efficiency, while the cost of H2 production plays a critical role in determining the economic viability of the coupled process. When the CO2 conversion rate increases from 47% to 79%, the energy efficiency of the coupling process reaches 40.92% and the total capital investment can be reduced by 38.79%. When the cost of H2 production decreases from 0.367 USD/m3 (standard conditions) to 0.114 USD/m3, the production cost (refers to “olefins production cost”) of the coupled process can be reduced by 51.58%, reaching as low as 1006.1 USD/t. At the similar H2 production cost (0.317 USD/m3), the production cost of the process is 2100.8 USD/t, which is lower than the reported value (3580 USD/t) in literature.关键词:CO2 hydrogenation;green olefins;Aspen Plus stimulation;process feasibility analysis;economic analysis100|0|0更新时间:2026-02-02 -
 摘要:NH3 is a crucial inorganic chemical raw material and an ideal hydrogen energy carrier. Ru-based catalysts have attracted significant attention due to their superior catalytic performances under low-temperature and low-pressure conditions compared to traditional iron-based catalysts. xCs-Ru/h-BN (x = n(Cs)/n(Ru)) catalysts were synthesized by supporting Ru and the promoter CsNO3 onto four commercial hexagonal boron nitride (h-BN) substrates. The physicochemical properties of the support and catalysts were characterized by XRD, SEM, N2 adsorption/desorption, H2-TPR, and XPS. The catalytic performances for ammonia synthesis of four catalysts under mild conditions were evaluated, and the reaction orders of N2, H2 and NH3 were analyzed through kinetics. The results show that the Ru species on the surface of 2Cs-Ru/h-BN-1 exhibits higher activity, effectively lowering the decomposition and reduction temperatures of CsNO3 and thereby facilitating N2 dissociation on the Ru active sites. Kinetic analysis reveals that the hydrogen reaction order of 2Cs-Ru/h-BN-1 is positive, indicating that the dissociated hydrogen atoms on the catalyst surface are more prone to migration and transformation. This not only suppresses hydrogen poisoning of the catalyst to stabilize ammonia synthesis but also releases more active sites for the dissociation of N2 molecules. Under the reaction conditions of 400 ℃, 0.1 MPa and syngas flow rate of 60 mL/min (V(H2):V(N2) = 3:1), 2Cs-Ru/h-BN-1 achieves an ammonia synthesis rate (calculated by per gram of catalyst) of 3902 μmol/(g·h) after 1 h of reaction and can operate stably for 48 h.关键词:hexagonal boron nitride;Cs-Ru/h-BN catalyst;ammonia synthesis;hydrogen poisoning4|0|0更新时间:2026-02-02
摘要:NH3 is a crucial inorganic chemical raw material and an ideal hydrogen energy carrier. Ru-based catalysts have attracted significant attention due to their superior catalytic performances under low-temperature and low-pressure conditions compared to traditional iron-based catalysts. xCs-Ru/h-BN (x = n(Cs)/n(Ru)) catalysts were synthesized by supporting Ru and the promoter CsNO3 onto four commercial hexagonal boron nitride (h-BN) substrates. The physicochemical properties of the support and catalysts were characterized by XRD, SEM, N2 adsorption/desorption, H2-TPR, and XPS. The catalytic performances for ammonia synthesis of four catalysts under mild conditions were evaluated, and the reaction orders of N2, H2 and NH3 were analyzed through kinetics. The results show that the Ru species on the surface of 2Cs-Ru/h-BN-1 exhibits higher activity, effectively lowering the decomposition and reduction temperatures of CsNO3 and thereby facilitating N2 dissociation on the Ru active sites. Kinetic analysis reveals that the hydrogen reaction order of 2Cs-Ru/h-BN-1 is positive, indicating that the dissociated hydrogen atoms on the catalyst surface are more prone to migration and transformation. This not only suppresses hydrogen poisoning of the catalyst to stabilize ammonia synthesis but also releases more active sites for the dissociation of N2 molecules. Under the reaction conditions of 400 ℃, 0.1 MPa and syngas flow rate of 60 mL/min (V(H2):V(N2) = 3:1), 2Cs-Ru/h-BN-1 achieves an ammonia synthesis rate (calculated by per gram of catalyst) of 3902 μmol/(g·h) after 1 h of reaction and can operate stably for 48 h.关键词:hexagonal boron nitride;Cs-Ru/h-BN catalyst;ammonia synthesis;hydrogen poisoning4|0|0更新时间:2026-02-02 -
Experimental study on corrosion behaviors of 310S stainless steel under high-temperature ammonia gas
 摘要:Due to the carbon-free and hydrogen-rich characteristics, ammonia gas is increasingly widely used in industrial and new energy fields. However, the ammonia gas corrosion problem in pipeline systems under high-temperature working conditions has become a key bottleneck restricting the development of the ammonia industry. 310S stainless steel was selected as the research object and a high-temperature ammonia corrosion experimental platform was built to simulate the actual conditions, and characterization techniques such as SEM, EDS and XRD were comprehensively applied to study the corrosion behaviors of 310S stainless steel under ammonia gas at different time and temperatures. The influence of tungsten carbide (WC) coating on corrosion resistance of 310S stainless steel was compared and analyzed. The results show that the annual corrosion rate of 310S stainless steel is relatively high in the early stage of corrosion and then gradually decreases. For example, when 310S stainless steel is corroded at 450 ℃ for 80 h and 480 h, its annual corrosion rates are 1.68 mm/a and 0.53 mm/a, respectively. The temperature has significant impact on the corrosion behaviors of 310S stainless steel. Among them, the corrosion is most intense at 450 ℃. For instance, when the corrosion time is 80 h, the annual corrosion rate of 310S stainless steel at 450 ℃ is 1.68 mm/a. It is significantly higher than its annual corrosion rate at 300 ℃ (0.79 mm/a) and 490 ℃ (1.09 mm/a). When the temperature is 450 ℃, the WC coating can effectively inhibit the penetration of active N into substrate of 310S stainless steel. However, at 490 ℃, the WC coating can experience local peeling, which can aggravate the corrosion of the substrate.关键词:310S stainless steel;high-temperature ammonia corrosion;tungsten carbide coating;corrosion behaviors157|0|0更新时间:2026-02-02
摘要:Due to the carbon-free and hydrogen-rich characteristics, ammonia gas is increasingly widely used in industrial and new energy fields. However, the ammonia gas corrosion problem in pipeline systems under high-temperature working conditions has become a key bottleneck restricting the development of the ammonia industry. 310S stainless steel was selected as the research object and a high-temperature ammonia corrosion experimental platform was built to simulate the actual conditions, and characterization techniques such as SEM, EDS and XRD were comprehensively applied to study the corrosion behaviors of 310S stainless steel under ammonia gas at different time and temperatures. The influence of tungsten carbide (WC) coating on corrosion resistance of 310S stainless steel was compared and analyzed. The results show that the annual corrosion rate of 310S stainless steel is relatively high in the early stage of corrosion and then gradually decreases. For example, when 310S stainless steel is corroded at 450 ℃ for 80 h and 480 h, its annual corrosion rates are 1.68 mm/a and 0.53 mm/a, respectively. The temperature has significant impact on the corrosion behaviors of 310S stainless steel. Among them, the corrosion is most intense at 450 ℃. For instance, when the corrosion time is 80 h, the annual corrosion rate of 310S stainless steel at 450 ℃ is 1.68 mm/a. It is significantly higher than its annual corrosion rate at 300 ℃ (0.79 mm/a) and 490 ℃ (1.09 mm/a). When the temperature is 450 ℃, the WC coating can effectively inhibit the penetration of active N into substrate of 310S stainless steel. However, at 490 ℃, the WC coating can experience local peeling, which can aggravate the corrosion of the substrate.关键词:310S stainless steel;high-temperature ammonia corrosion;tungsten carbide coating;corrosion behaviors157|0|0更新时间:2026-02-02
0

